Semi-continuous bioprocess for whole cut cultivated meat using simultaneous perfusion and stretch
Stretching of engineered muscle constructs has been previously demonstrated to induce alignment and maturation of muscle fibers, which is desirable for whole cut cultivated meat. Stretch stimuli could also be incorporated into a semi-continuous bioprocess in which a piece of tissue is expanded over time and portions of the tissue periodically harvested. The large amount of meat produced could offset the high initial cost of fabricating a construct capable of continuous growth.
-
Cultivated
- Research
- Commercial
- Production
- R&D
- Bioprocess design
- Scaffolding
- Industry
- Academics
- Startups
Current challenge
When choosing a tissue engineering strategy, a trade-off exists between faithfully mimicking the complexities of naturally-occurring muscle tissue and ensuring that the selected approach is sufficiently scalable and economical for large-scale manufacturing of the variety of structured products pursued by the CM industry. For example, in 3D printing, speed and resolution tend to have an inverse relationship. Much of the complexity associated with biologically functional tissues is likely unnecessary to replicate—a piece of CM that is acceptable to even the most discerning food critic might be easily distinguished from conventional meat under a microscope. However, only small samples of CM have thus far been produced outside of for-profit companies. Therefore, the production methods under investigation by academic researchers have not fully been put to the test regarding either their sensory acceptability for creating whole-cut products or the realities of how easily they will scale. Thus, additional innovations may be needed to produce cultivated meat that is simultaneously delicious and cost-effective.
Proposed solution
One possible strategy which, to our knowledge, has not been investigated is the engineering of a highly detailed tissue construct through high-resolution 3D bioprinting or another method, followed by the application of a gradual stretch stimulus in a perfusion bioreactor (see figure a). At the end of the growth period, the center of the construct could be gradually constricted and then cut to harvest a portion of the tissue. The cycle would then repeat with the portion remaining in the bioreactor. There is precedent for the idea of expanding a dense and complex muscle tissue in culture. One of the earliest studies on CM demonstrated an increase in the size of goldfish muscle explants when they were cultured with brown bullhead fibroblasts. The stretch stimulus would be expected to promote further growth since previous studies (Costa et al. 2003, Phillips et al. 2014, Puetzer et al. 2015, Thomopoulos et al. 2005, Agrawal et al. 2017, Bowles et al. 2010, Vandenburgh & Karlisch 1989) have reported effects of passive stretch on myotube alignment and maturation and on cell proliferation. Several stretch regimes are possible (see figure b) during the growth/constriction phase, including a simple continuous stretch, a phasic stretch where the length of the construct is held constant during the relaxation periods, or a phasic stretch where the construct is allowed to shorten between stretches (Okano & Matsuda 1998, Fink et al. 2000). The effects of such regimes would need to be tested empirically and balanced against differences in energy consumption. For large-scale production, such a process could be partially or fully automated, and in-line sensors could be used to dynamically adjust the growth period and culture conditions.
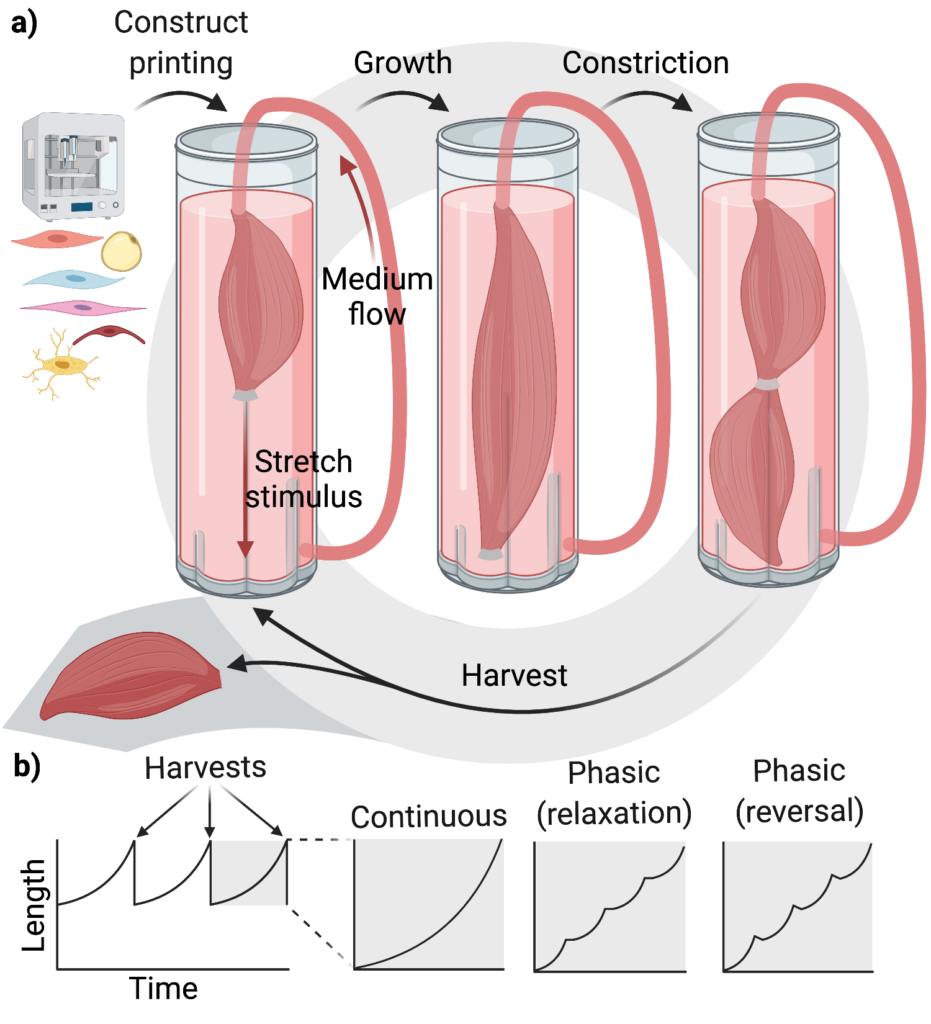
b) Length of the hypothetical construct over time. Insets indicate three possible forms the stretch stimulus could take within each growth phase.
Other strategies for the initial fabrication step that might be lower in cost are the use of endothelialized spinach leaves, other plant material, or organoid-based constructs. The self-organizing properties of the cells in such constructs might be sufficient to achieve the correct muscle-like 3D organization. Periodic additions of fresh satellite cells, myoblasts, or preadipocytes to the construct could further stimulate growth if necessary.
Anticipated impact
With the right combination of proliferative adult stem cell types, a functional vascular network capable of producing new capillaries as needed, and the right culture conditions, a construct subjected to long-term stretch and perfusion might be capable of continuous growth. While the initial fabrication of such a construct would otherwise almost certainly be cost-prohibitive, and the level of complexity likely unnecessary from an organoleptic perspective, continuous growth would allow for multiple harvests. Given a reliably sterile environment, growth could potentially continue over a long period. It might also be possible to use the harvested tissue to seed additional bioreactors, thereby reducing the cost of ramping up to large-scale production. Such constructs could be fabricated with large-scale heterogeneity designed to mimic the different cuts of meat produced in vivo.
Related efforts
Several academic research groups, including several supported by GFI, are working on bioreactor and bioprocess design innovations. Passive stretch (via anchoring of construct ends) was used by Kang et al. (2020), Simsa et al. (2019), and Furuhashi et al. (2021) to promote alignment and maturation in small-scale cultivated meat prototypes. Mark Post’s group also employed passive stretch in the form of constrained ring-shaped constructs to produce the first cultivated meat burger in 2013.
GFI resources

Research funding database
GFI’s research funding database provides curated grant opportunities for open-access alternative protein research.

Collaborative Researcher Directory
Use this directory to find scientific collaborators in the alternative protein field.

Find collaborators
Join the GFIdeas global community of 2,000+ entrepreneurs, scientists, investors, and subject matter experts. Discuss projects on the members-only Slack community, attend monthly seminars, and use the community directory to help you find collaborators working on similar Solutions!
Related solutions
-
Cultivated
Computational models of perfusion flow through scaffolds
For tissue-structured cultivated meat production, the transition from the proliferation phase to differentiation phase may involve seeding cells onto a prefabricated scaffold within a perfusion bioreactor. Medium is then perfused…
-
Cultivated
3D microenvironments for cell expansion
Proliferation and high-density cell growth are fundamentally important to scaling cultivated meat production. Recent demonstrations of stem cell expansion in 3D microenvironments such as encapsulated spheres or tubules can generate…
-
Cultivated
Post-harvest processes and end product characterization for cultivated meat
A number of cellular processes occurring after slaughter are known to affect the quality and sensory properties of conventional meat. Cultivated meat will offer unprecedented control over these parameters and…

Explore the full solutions database
Browse 100+ startup ideas, commercial opportunities, research projects, and investment priorities throughout the alternative protein supply chain.
Get involved
If you’d like to fund a research project, work on any of these solutions, share information about related efforts that are already underway, or elevate new ideas for advancing the alternative protein industry, we’d love to hear from you!
