
Deep dive: Fermentation protein ingredients and food functionality
Explore the functionality of fermentation-derived food ingredients conferred by the production method, preparation, and microbe.
Overview of fermentation-derived protein ingredients
By 2050, the global population is projected to reach 9.7 billion, significantly increasing demand for essential macronutrients, particularly protein. Protein is vital for maintaining physiological functions and metabolic balance in the human body, and its deficiency can lead to hormonal imbalances and inefficient energy metabolism. (B. Wang et al. 2023)
The growing challenges and impact of our current agricultural system have increased interest in alternative protein sources for food (Poore and Nemechek 2019). Fermentation-derived (FD) alternative proteins (AP), typically referred to as mycoprotein or single-cell proteins (SCP), are a promising solution to global protein shortages, offering significant environmental and nutritional advantages. FD AP production offers notable advantages over conventional protein sources, including shorter production cycles, reduced land requirements, and suitability for diverse climates. New advances in food science and production are demonstrating the benefits of FD proteins, such as high protein, vitamin, and lipid content, and overcoming historical challenges in protein ingredient functionality, high nucleic acid content, low digestibility, and limited availability (Ugbogu and Ugbogu 2016). Progress over the past decade has led to considerable advances in the compositional and functional attributes of fermentation-derived alternative proteins.
Research and development in alternative proteins have driven rapid innovation. These products are increasingly appealing to mainstream consumers, particularly flexitarians. Over the past decade, the food industry has seen evolving demand for diverse protein sources. The development of alternative proteins through microbial fermentation has introduced two emerging industries in food production: precision fermentation (PF), which uses microbes to produce specific animal proteins, and fermentation to produce high-protein microbial biomass. Biomass proteins are classified as single-cell protein (SCP) when derived from yeast/bacteria/microalgae. Protein ingredients produced by filamentous or mycelial fungi are often called mycoprotein. These innovations in food technology and agriculture continue to significantly widen their market presence and organoleptic appeal (Hefferon et al. 2023).
Fermentation has been used since ancient times for food preservation and has evolved into a diverse process for producing various fermented foods and beverages from many feedstuffs. Currently, three types of fermentation are available for food applications:
- Traditional fermentation produces foods and beverages using microorganisms like bacteria and yeasts, which may be naturally present or added as starter cultures. This process enhances the sensory and nutritional properties of food products by modifying their organoleptic qualities. While interest in traditional fermentation is resurgent, this deep dive will not extensively cover the functionality of food ingredients related to traditional fermentation.
- Biomass fermentation focuses on producing mycoprotein and SCP as high-protein ingredients that also provide other nutrients. Sometimes, biomass fermentation approaches use sidestreams such as food processing by-products and agricultural residues as substrates/feedstocks, an environmentally friendly and efficient approach for sustainable food production.
- Precision fermentation uses microbes to produce specific high-value molecules, such as proteins, pigments, vitamins, and fats. These molecules are biomanufactured and prepared as ingredients in food products like alternative meats and dairy. (Boukid et al. 2023).
Biomass-derived (BD) fermentation products have been commercially available for several decades, providing protein ingredients for center-of-plate products and other applications where functional proteins enhance end products. Precision fermentation (PF) for food ingredients represents a recent technological breakthrough, offering modeled resource efficiencies superior to traditional livestock farming (Knychala et al. 2024). Despite its seemingly recent application to food, PF approaches have been used for decades to produce food preparation enzymes like rennet and dough conditioners. With its potential to revolutionize food systems, PF focuses on producing milk proteins, egg proteins, and specific components for alternative meat products, enabling more sustainable and potentially localized food production (Nielsen, Meyer, and Arnau 2024).
Despite these advancements, we must address critical research gaps and challenges to realize the full potential of FD proteins in food applications. Key areas requiring attention include:
- Optimizing production efficiency to reduce costs and scale manufacturing for global markets.
- Enhancing sensory and functional properties, such as taste, texture, and aroma, to increase consumer acceptance of FD proteins.
- Navigating complex regulatory frameworks to ensure food safety and quality while fostering public trust.
In particular, understanding the functional behaviors of FD proteins — such as solubility (the ability to dissolve in water), emulsification (stabilizing oil-water mixtures), foaming (forming and stabilizing air bubbles), gelation (creating protein networks for texture), and water-holding capacity (retaining moisture for juiciness and texture) — is critical for replicating the qualities of animal-derived proteins in alternative products. Sensory properties, on the other hand, encompass the attributes perceived by the senses, such as taste (basic flavors like sweet, salty, sour, and bitter), texture (mouthfeel like hardness, cohesiveness, springiness, gumminess, and chewiness), aroma (smell that influences flavor perception), and appearance (visual elements like color and sheen). Enhancing these properties in alternative proteins is crucial to replicating the organoleptic appeal and functionality of animal-derived products, thereby improving consumer acceptance and overcoming barriers to adoption. Furthermore, these efforts will be pivotal for producing appealing dairy, egg, and meat substitutes. To meet consumer expectations, future innovations must focus on production methods, sensory attributes, and functionality to enhance these alternatives to broader markets.
This review provides a comprehensive overview of FD proteins for food applications, focusing on their microbial hosts, innovations, challenges, and opportunities in alternative meat, egg, and dairy production. Commercial innovation in FD APs began in the 1980s with BD products, whereas the commercialization of PF protein ingredients occurred far more recently. Therefore, much of the information summarized and reported here focuses on BD ingredients and their functionality.
Fermentation-derived protein production microbes
FD proteins rely on microbial hosts such as yeasts, filamentous fungi, bacteria, and algae for large-scale production. Specific microorganisms are selected for their ability to grow rapidly, utilize diverse substrates, and produce safe and high-quality proteins suitable for food applications. Emerging technologies, such as hybrid microbial systems, are also being explored to combine the strengths of different microbial hosts, offering enhanced functionality and scalability. In the following sections, we will explore these microbial hosts in detail, focusing on their unique characteristics, advantages, and applications in the production of alternative proteins.
Biomass-derived protein production
Microbe-derived proteins are typically produced by growing food-safe microorganisms in bioreactors or controlled environments during fermentation. The fermentation-derived biomass is harvested via centrifugation or filtration and can undergo protein extraction or further processing. While dry biomass exhibits reduced oil-binding and foaming capabilities due to its high insoluble fiber content, extracted proteins demonstrate improved techno-functional properties such as solubility, emulsification, and gelation capacity. Centrifuged/de-watered fungal biomass naturally resembles ground meat in texture (Malila et al. 2024). This biomass can be dried and processed into chunks, blended into ground meat or comminuted products, or formed into whole-muscle meat using binding agents (Malila et al. 2024). Other microbial biomass, such as yeast and microalgae, can produce protein-rich flours when dried and can be formulated and texturized into products using processes similar to those for plant-based proteins, such as extrusion. See GFI’s plant-based meat end product formulation and manufacturing deep dive for more.
As shown in Figure 1A, microbial biomass-derived protein production can involve various fermentation processes (e.g., submerged, semi-solid, or solid-state fermentation) depending on the source organism (bacteria, yeasts, fungi, or algae). Key steps include fermentation, RNA reduction, filtration, purification, and subsequent processing such as drying or chilling. These processes are tailored to optimize protein yield and quality for specific applications, such as microbial proteins, mycoproteins, or algae-derived proteins.Recent advancements in formulation and texturization, such as food 3D printing, have enhanced the versatility of microbial FD ingredient proteins, enabling innovations in textures, colors, shapes, and sizes of food products (Figure 1B). These developments support the increasing use of biomass-derived protein in alternative proteins and diverse food applications. Figure 1B highlights downstream processing technologies for converting microbial proteins into food products. These include extrusion, spinning techniques, shear cell technology, and innovative approaches such as 3D printing. Adding binding agents and cross-linkers before these processes, including polysaccharides, proteins, or enzymes, can transform ingredient texture to enhance the final products’ structural and functional properties and appeal. Auxiliary technologies like spray-drying, freeze-drying, and microwave heating play a critical role in preserving and stabilizing these proteins. Together, these methods enable the creation of diverse food products with various textures, shapes, colors, and applications, showcasing the versatility of microbial proteins in modern food systems (Maini Rekdal et al. 2024).
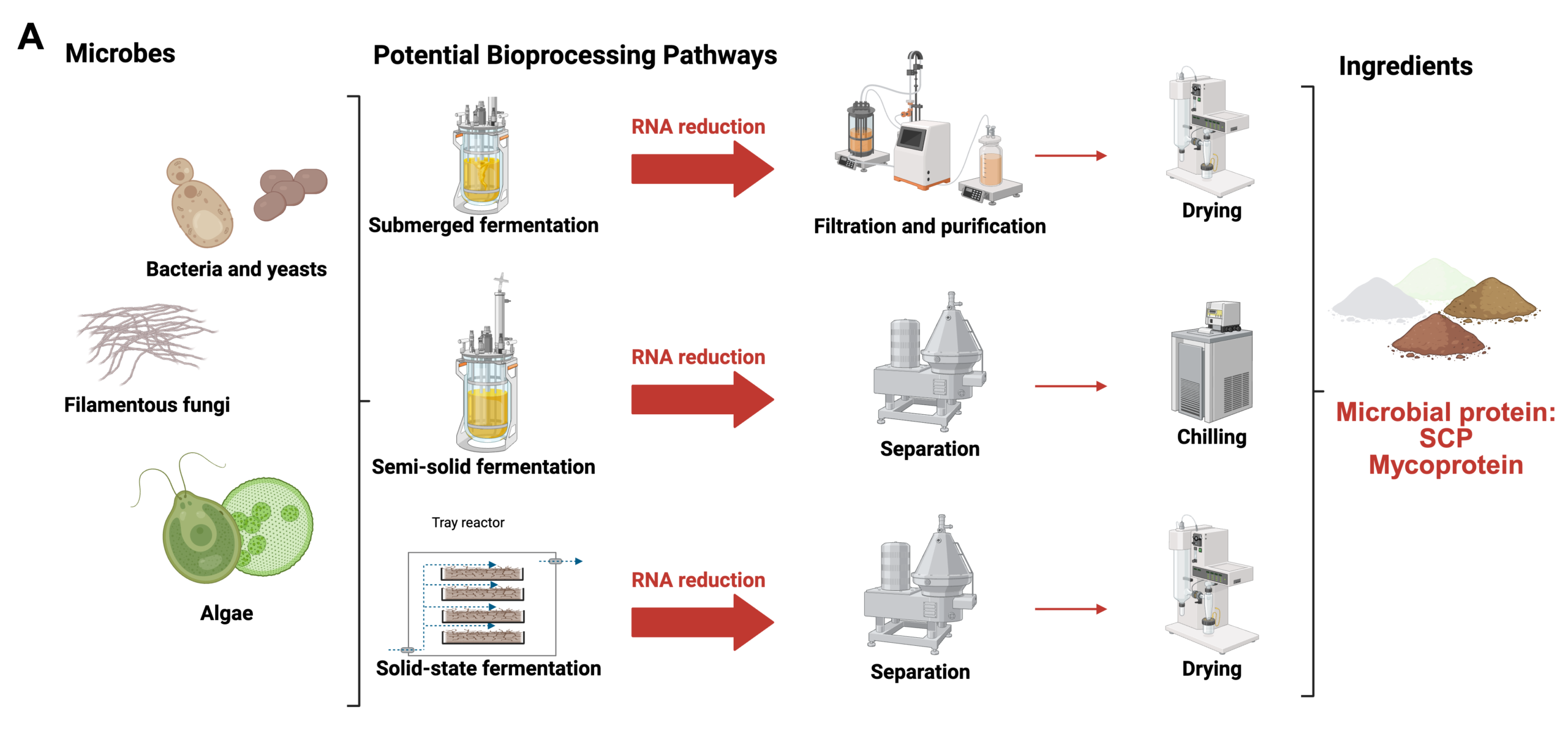
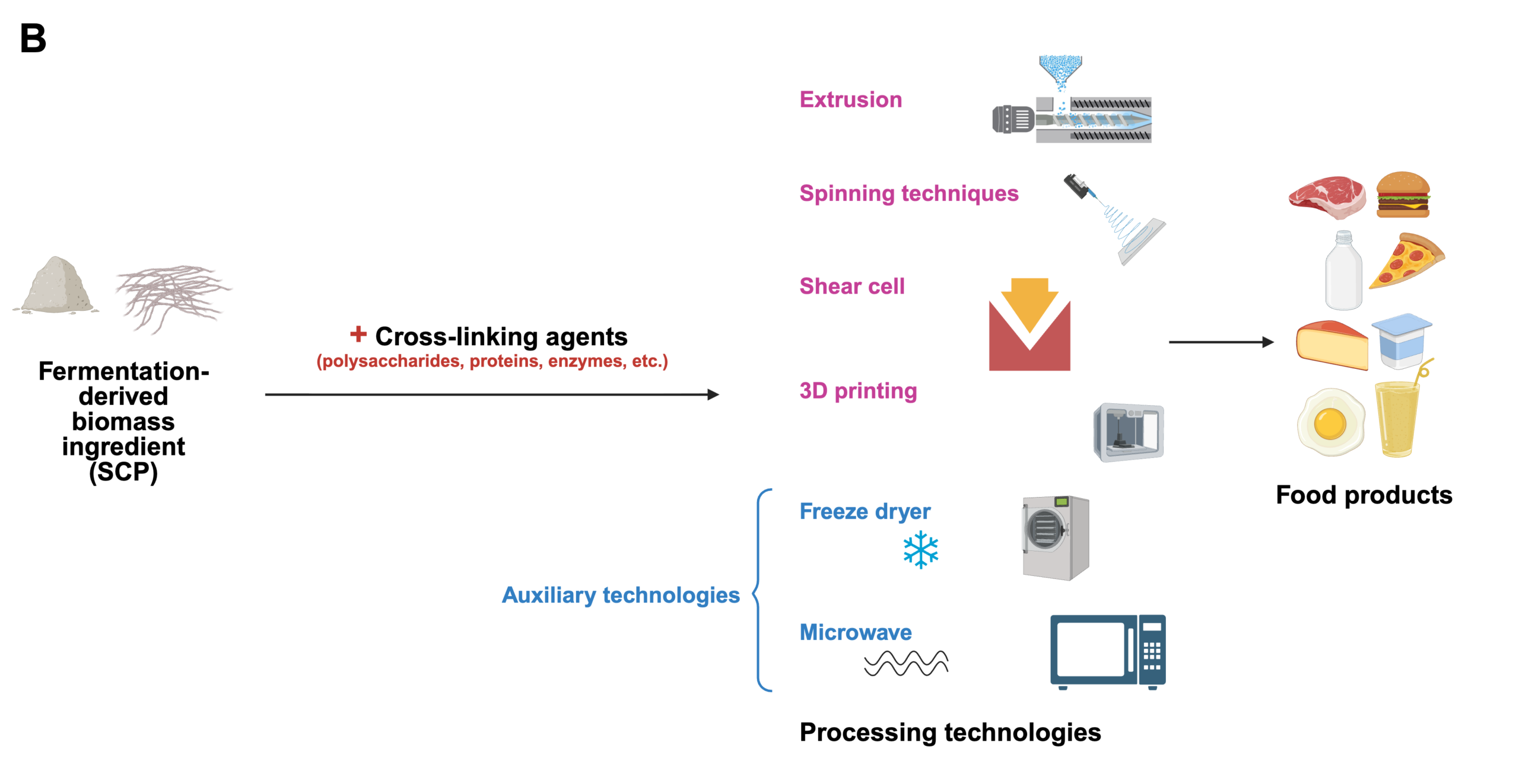
Figure 1. A) Diagram of the Production Process for Microbe-Derived Alternative Proteins. B) Formulation and texturization of microbial-based food products (Barzee et al. 2021). Created with BioRender.com.
Precision fermentation-derived protein production
The production of specific proteins by PF shares many aspects with BD protein production, namely growing microbes in a bioreactor using carbon and nitrogen feedstocks. However, rather than consume the feedstock materials to accumulate high-protein biomass, cells are induced to produce large quantities of a particular protein. The production of the target protein is typically made possible through engineering the microbe (Figure 2). Molecular biology approaches guide the target gene to the host microbe genome or a plasmid under the control of a promoter that enables high expression of the RNA for the target gene. Subsequently, ribosomes translate the expressed RNA into the protein of interest. Downstream processing, the steps required for isolation and purification of the target protein, is more complex in PF. Typically, this process involves separating the target protein from other cellular material, like lipids and carbohydrates, then isolating it from other host microbe proteins. Isolation and purification of the target proteins by size, charge, and/or solubility leads to a prepared ingredient meeting the specifications required for food use (Eastham and Leman 2024). Depending on the food application and storage requirements, the protein can be dried or prepared as a solution in water.
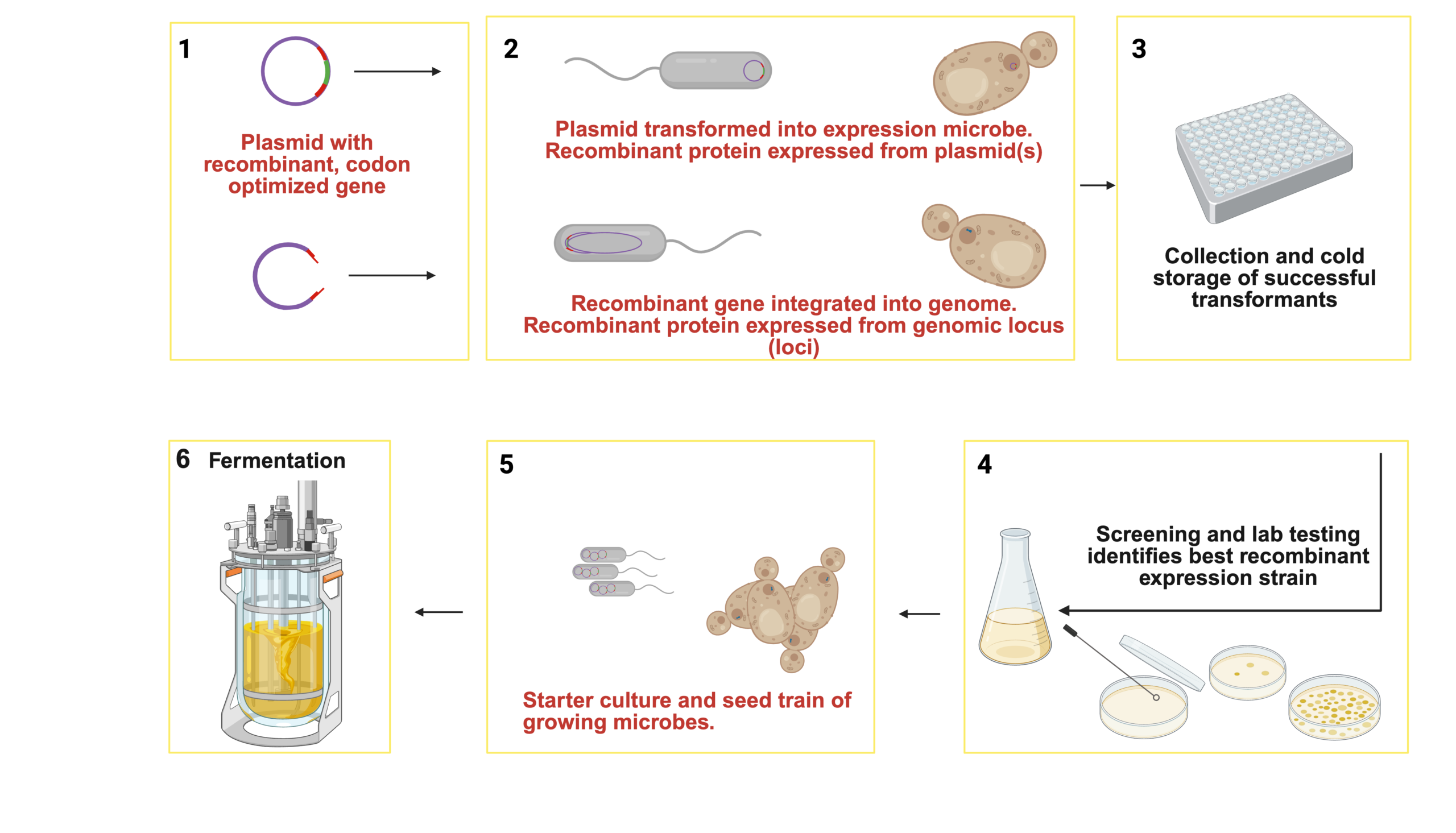
Figure 2. The production of recombinant proteins involves isolating a gene, codon optimizing it for the expression micros, inserting it into a vector, such as a plasmid (1). The modified vector is introduced into a host cell (e.g. bacteria, yeast), where it can stably maintain as an extrachromosomal plasmid or integrate the recombinant gene cassette into the genome (2). Successfully transformed cells are identified and stored (3). The candidate strains are screened for recombinant protein expression (and other bioprocess-relevant attributes (4). The strain is inoculated into growth medium and progressively scaled in fermentation (5,6). Later, successful process relies on optimized fermentation conditions and efficient downstream processing to produce and purify the desired recombinant protein (Diaz-Bustamante et al. 2023). Created with https://BioRender.com.
Yeast fungi
Budding yeasts are highly effective for fermentation, offering scalability, rapid growth, and protein-folding capabilities similar to animal cells (Eastham and Leman 2024). Among the 1,500 known yeast species, common food-safe yeasts include Saccharomyces cerevisiae, Yarrowia lipolytica, Candida species, Komagataella phaffii, and Kluyveromyces species (B. Wang et al. 2023; Eastham and Leman 2024). S. cerevisiae, due to its low nucleic acid content, is a long-standing and cost-effective microbe for single-cell protein (SCP) production. It is generally recognized as safe (GRAS) and free from major allergenic risk, making it an excellent protein source for human and animal nutrition (B. Wang et al. 2023). However, for PF applications, its split aerobic/anaerobic metabolism, high ethanol production, and hyperglycosylation of some proteins can make it less favorable (Eastham and Leman 2024). Komagataella phaffii is an example of a preferred PF microbe, due to its high secretory efficiency and proven success in producing proteins like lactoferrin and myoglobin. Similarly, Kluyveromyces species are promising due to their high protein secretion and enzyme production. For more information on yeast PF strains, see GFI’s mini-review of PF proteins (Eastham and Leman 2024).
Yeast biomass-derived proteins, comprising 40–60% protein by dry weight, are highly nutritious and comparable to meat and soybeans, with protein concentrations exceeding those of dairy milk. These complete proteins contain all 20 essential and non-essential amino acids and are rich in flavor-enhancing amino acids like glutamic acid and lysine. However, sulfur-containing amino acids, such as methionine and cysteine, can be present in lower amounts in yeast biomass-derived proteins, which could limit their standalone nutritional adequacy for some applications. Yeast proteins are highly bioavailable, with 96% digestibility and a higher protein efficiency ratio (PER) than conventional proteins. Despite these benefits, the high nucleic acid content and production cost hinder their commercial use (B. Wang et al. 2023). In addition to their nutritional value, yeast proteins have desirable functional properties for food formulations, including solubility, flavor enhancement, nutritional supplementation, foaming, and emulsification. Expanding on these functional attributes, yeast proteins exhibit excellent functional properties, such as solubility, water-holding capacity (WHC), emulsification, and oil-holding capacity, making them versatile for various food applications, including their use as meat extenders. Their high solubility and WHC contribute to improved texture, dimensional stability, and cooking performance in meat products like sausages, burger patties, and deli meat.
Additionally, yeast proteins offer a low-risk alternative to traditional meat extenders such as textured soy protein (TSP) or flours, which may pose an allergy risk for some individuals, thereby broadening their consumer appeal. However, their functionality varies depending on yeast species, extraction conditions, and processing methods. For instance, high-pressure homogenization or the use of salt may alter solubility and WHC. While bitterness and undesirable flavors caused by compounds like iso-α-acids and tannins can present sensory challenges, pre-debittering treatments can effectively improve sensory acceptability without compromising functionality. Despite these challenges, yeast proteins hold significant potential as cost-effective, sustainable protein ingredients that address global protein shortages and improve the nutritional profile of meat products (Ma et al. 2023).
For instance, (Jeon, Gu, and Ryu 2022) investigated yeast integration into low-moisture extruded meat analogs, assessing its impact on physicochemical properties such as texture, water-holding capacity, and protein solubility. The study used a base formulation of soy protein, wheat gluten, and cornstarch with yeast inclusion levels ranging from 0% to 50%, processed under specific extrusion conditions. The results revealed that yeast content significantly influenced properties like texture and antioxidant activity, with 30% yeast inclusion identified as optimal for achieving desired texturization and functionality in alternative meat products. The findings highlight the potential of yeast as a functional and structural ingredient in developing hybrid plant-based and FD alternative meat products (Jeon, Gu, and Ryu 2022).
Beyond alternative meat applications, microbial SCPs can contribute to diverse food categories, including dairy and bakery products. Rheological analyses indicate that SCPs derived from kefir microorganisms, such as yeasts and lactic acid bacteria, produce gels with a firmer texture than those made with soy flour, likely due to their high protein content (Paraskevopoulou et al. 2003). Additionally, Solein®, a bacterial SCP developed by Solar Foods, exhibits strong water- and oil-binding properties and foaming and emulsifying capacities compared to pea protein isolate. Similar attributes make bacterial SCPs promising functional ingredients for use in baked goods, pasta, yogurt, or microbial-based meat products (Molfetta et al. 2022).
Generally, these results underscore the suitability of microbial biomass for applications in baked goods, snacks, and alternative meat products. However, research into the functional properties of yeast proteins remains early, with most studies focusing on generic yeast proteins without detailing species-specific characteristics, growth conditions, or proteome makeup characteristics. The variability in amino acid composition, solubility, and protein structure across different yeast species and fermentation conditions presents challenges and opportunities for optimizing yeast biomass for specific food applications (Martin and Chan 2024). Advances in understanding yeast metabolism and refining bioprocessing techniques allow for better control of yeast proteomes to meet the demands of scalable and economic food production. Computational methods have introduced innovative approaches to optimize yeast metabolism for protein production, including analyzing and selecting microorganisms with desirable nutritional attributes to enhance protein yield and quality (Zielinski et al. 2020; Karlson et al. 2023; Vinestock et al. 2024). In practice, engineered metabolic pathways and targeted bioprocess adjustments improve protein functionality and amino acid content while maintaining cost-efficiency and environmental sustainability (Brubaker et al. 2023). Future innovations will focus on reducing production costs, utilizing sustainable feedstocks, and optimizing bioprocessing techniques to develop yeast proteins with desirable nutritional and functional attributes.
Filamentous fungi
Several filamentous fungi, such as Fusarium venenatum, Neurospora intermedia, and Aspergillus, are common and established in food applications. Filamentous fungi are morphologically distinct, with long projections called hyphae used to explore their environment and accumulate nutrients. Filamentous fungi, traditionally used in producing foods like koji, tempeh, miso, and sake, offer significant opportunities for meat alternatives. Traditional foods utilize strains such as Aspergillus (koji) and Rhizopus (tempeh), and their food safety profiles, growth characteristics, and protein content have made them attractive for alternative protein applications. At the same time, more recently identified molds like Fusarium species have demonstrated high potential in modern food applications. For example, F. graminearum, later identified as F. venenatum, demonstrated high protein yield, low odor, and minimal toxicity, making it ideal for fermenter-based growth. In 1985, F. venenatum became the basis for the first commercial mycoprotein product, Quorn. Since then, mycoprotein has gained widespread attention due to its environmental benefits, commercial viability, and center-of-plate alternative meat products (B. Wang et al. 2023).
Fungi play a pivotal role in the food fermentation industry, and the growth of the FD AP industry has encouraged a renewed focus on the nutritional profiles of filamentous fungi. Fungal proteins, comprising 45%–54% of their dry weight, exhibit essential amino acid profiles comparable to yeast proteins (B. Wang et al. 2023). They also have low fat levels, no cholesterol (Li et al. 2023), and a high protein digestibility-corrected amino acid score (PDCAAS) similar to milk protein and higher than chicken and beef. Studies demonstrate that fungal protein consumption promotes muscle protein synthesis and sustains plasma amino acid and insulin levels in healthy adults (B. Wang et al. 2023; West et al. 2022; Hannanian and Churchward-Venne 2023).
In addition to these nutritional benefits, fungal proteins enhance gut health by promoting beneficial gut microbiota, reducing fecal genotoxicity, and potentially preventing colorectal diseases (Colosimo et al. 2024; Farsi et al. 2023). For example, β-glucan from fungal cell walls has been linked to lower cholesterol and lipid levels. At the same time, immunomodulatory proteins (Fungal Immunomodulator Proteins – FIPs) exhibit anti-inflammatory and potential anti-cancer properties (Li et al. 2023).
Beyond their nutritional value, fungal proteins exhibit functional benefits that improve flavor in food products. Fungal strains, culture medium, and processing methods influence their umami taste and meat-like texture, with some fungi imparting natural umami or earthy notes. In contrast, others with neutral flavor may require flavor additives like spices or extracts. Texture and protein binding are often accomplished through additives during formulation, such as albumin protein in some Quorn™ formulations, or in post-cultivation processing like extrusion and pressing.
Fungal protein functional benefits are influenced by fungal morphology and growth conditions. For example, smaller harvested yields from bioreactors typically experience higher oxygen transfer, which can lead to denser biomass (Ogawa et al. 2024). The color of fungal food products can also be modified during or after cultivation. Fungal cells can adsorb pigments from their growth media, enabling natural or artificial coloring. Using fungus’s endogenous pigments and external colorant sources, formulators can fine tune the product color and appearance. Edible fungi can also be engineered for enhanced sensory and nutritional characteristics, expressing nutritionally beneficial molecules like iron-binding heme (Maini Rekdal et al. 2024). The microbe’s absorption or accumulation of certain molecules can also improve micronutrient profiles. Coculture fermentation with other microorganisms can positively influence the micronutrients and biomolecules present in the cell biomass, creating products with enhanced nutritional and biological performance (Barzee et al. 2021).
Fungal proteins are structurally similar to muscle fibers due to their filamentous structure, creating meat-like textures for alternative meat products like patties, sausages, and ground orminced meat. Mushrooms or edible macrofungi such as Agaricus bisporus, Pleurotus sp., Shiitake sp., and Flammulina velutipes are widely cultivated using agricultural residues for sustainable production. The mycelium of these mushroom species are being investigated and commercialized for FD protein production. Additionally, fungal strains like Penicillium roquefortii and Aspergillus oryzae contribute to cheeses and other traditional fermented products (Li et al. 2023). These species are of commercial and food science interest for their sensory molecules and biomass production. Filamentous fungi are also popular and highly efficient PF production platforms, producing large quantities of recombinant proteins and enzymes like amylases and cellulases, with species such as Aspergillus and Trichoderma reesei achieving high protein titers. However, challenges like culture viscosity and long fermentation cycle durations remain. While titers for food proteins like ovalbumin and β-lactoglobulin are improving, further optimization is needed to meet commercial requirements (Eastham and Leman 2024). With roles in both biomass fermentation/SCPs and PF, filamentous fungi-based proteins have a prominent role in addressing global protein demands.
Bacteria
Bacteria are highly efficient hosts for FD proteins due to their rapid growth, scalability, and genetic flexibility. While Gram-negative bacteria like Escherichia coli can express PF-derived proteins such as collagen and Chymosin-B, their endogenous endotoxins can present challenges for strain engineering and bioprocess optimization. Further, E. coli is widely regarded for recombinant protein production due to its well-understood genetics and efficiency in protein expression, it is less common in food applications due to low protein titers and limitations in producing proteins that require specific PTMs for functionality. To address these issues, Gram-positive bacteria, including Bacillus subtilis, Corynebacterium glutamicum, and Lactococcus lactis, are sometimes preferred for their lack of endotoxins, efficient protein secretion pathways, and GRAS status. Strains of these species have successfully produced precommercial volumes of recombinant casein and heme proteins. Despite post-translational modification (PTM) and downstream processing challenges, bacterial PF offers competitive production costs for mid-to-high-value food proteins (Eastham and Leman 2024).
Microorganisms with GRAS status or non-pathogenic classifications remain the preferred choice for food applications, with PF often relying on safe and efficient hosts such as Bacillus species (Augustin et al. 2024). Advances in fermentation processes design and strain engineering have improved its efficiency, but its role in food protein production remains secondary to other hosts like yeasts and filamentous fungi (Knychala et al. 2024; Gupta and Shukla 2016).
Bacillus subtilis is well-suited for food-related recombinant protein production due to its non-pathogenic nature, high-density fermentation capacity, and efficient protein secretion pathways. Advances in fermentation techniques and process optimization have significantly enhanced its protein expression and secretion capabilities. These developments highlight the potential of B. subtilis as a robust and scalable platform for producing proteins essential for alternative food applications (Zhang, Su, and Wu 2020).
Bacterial SCPs, such as those produced by Cupriavidus necator and Xanthobacter sp., are gaining attention as sustainable, high-quality protein sources (Vlaeminck et al. 2023; Fasihi et al. 2025). Similarly, bacterial species such as Clostridium tyrobutyricum can be grown anaerobically to achieve high-protein biomass (Jonaitis et al. 2022). These bacterial protein flours are sometimes derived from renewable substrates like carbon gases and are rich in essential amino acids, offering functional properties such as water-binding and emulsifying capacities. Like yeast and fungi-based SCPs, bacterial proteins provide a sustainable alternative with growing potential in the food industry. Lactic acid bacteria (LAB) have enhanced fermented dairy products’ shelf life, flavor, and texture for millennia. They are now being applied to plant-based foods for similar improvements, offering nutritional benefits such as producing bioactive compounds, enriching the protein quality, and improving digestibility. Bacillus subtilis var. natto, traditionally used in Japanese natto production, also shows potential for plant-based fermentations, with legumes serving as suitable substrates (Rocchi et al. 2024), perhaps paving the way for its use in biomass applications. Combining LAB with non-sporulating B. subtilis strains improves texture, acidification, and overall nutritional quality in plant-based matrices, making them effective for creating alternatives like yogurt and sour cream. These advancements highlight the versatility of LAB and B. subtilis in enhancing plant-based food applications (Fernández-Varela et al. 2024).
Microalgae
Microalgae are photosynthetic organisms that offer another source of SCP. They contribute to food security and sustainability while providing slightly different food functionality roles from those of yeast and bacteria. In the wild, microalgae utilize carbon dioxide as their primary carbon source. Atmospheric carbon dioxide is sometimes supplemented in production settings with emissions from combustion, carbonates, or bicarbonates to enhance growth. Microalgae can also be grown heterotrophically using sugar sources (Ugbogu and Ugbogu 2016), and/or sunlight, carbon dioxide, sea or freshwater, and other readily available inputs, offering a sustainable feedstock for food, feed, energy, and biochemicals. Their rapid growth rates and higher productivity compared to traditional crops, combined with their ability to avoid competing for arable land, position them as a promising alternative protein source (Barbosa et al. 2023). Microalgae cultivation modes vary based on metabolic pathways:
- Photoautotrophic cultivation relies on light energy and carbon dioxide, releasing oxygen as a by-product.
- Heterotrophic cultivation uses organic carbon sources, such as sugars, in the absence of light.
- The mixotrophic approach combines both methods, optimizing growth and nutrient uptake by providing light/carbon dioxide and organic carbon as feedstock options.
Light availability is critical for phototrophic and mixotrophic growth, with three operational modes: sunlight, artificial light, or a combination of both. Sunlight is the most cost-effective option, but it is limited in regions with seasonal light constraints, such as the extreme northern and southern latitudes. Artificial light, although costlier, ensures consistent output, higher biomass yield, and stable quality, making it suitable for high-value microalgal products such as nutraceuticals and pharmaceuticals. For large-scale production of bulk microalgal products, direct sunlight remains the most economical feedstock choice, despite significant technical and process control challenges (Barbosa et al. 2023).
Algae, including cyanobacteria, microalgae, and seaweeds, are nutritionally diverse, with their composition varying by species, region, and environmental factors (Wu et al. 2023). Protein content in microalgae ranges from 3 to 70% of dry weight, depending on the species and growth conditions. For instance, the filamentous cyanobacteria Spirulina (Arthospira platensis) can reach up to 70% protein, surpassing many terrestrial plants such as legumes, cereals, and some traditional animal protein sources like milk. Algal proteins provide all nine essential amino acids, comparable to animal-based proteins like eggs, though they are lower in sulfur-containing amino acids such as cysteine and methionine. Also, algae are rich in glutamic acid, aspartic acid, alanine, and glycine, enhancing umami flavor and offering physiological benefits. These attributes position algae as a sustainable, nutritionally robust protein source, especially for plant-based diets (Wu et al. 2023).
In addition to their high protein content, microalgae provide functional benefits, such as stability and emulsifying properties similar to traditional food emulsifiers. Challenges remain, including optimizing large-scale protein extraction methods and addressing sensory attributes, such as green or dark pigmentation and earthy or fishy flavors, which can hinder consumer acceptability. Deodorization techniques, selective strain cultivation, and controlled growth conditions can mitigate these issues (Espinosa-Ramírez et al. 2023; Costa et al. 2024). However, microalgae flavors, colors, and textures can improve alternative seafood formulations (Coleman et al. 2022; Zhao, Khang, and Du 2024). Check out GFI grantee Dr. Jing Zhao’s research exploring this potential.
Building on these functional benefits, microalgal proteins are gaining attention for their versatility, excellent nutritional profile, and functional properties, making them suitable as binders, fillers, and flavor enhancers in alternative meat formulations. Algae-derived ingredients like Chlorella and Spirulina enhance textures, tenderness, and nutritional value, such as increased levels of vitamins B and E. Innovations include red seaweed for vegan bacon and algal biomass like “Essential Red,” which is rich in heme and used in alternative pork and meatball formulations. Commercial microalgae SCP producers have used tailored algal strains to produce high-protein biomass for plant-based and cultivated meats (Wu et al. 2023). Moreover, hydrocolloids derived from algae, such as carrageenan, agar, and alginates, provide essential gelling, thickening, and stabilizing properties, ensuring cohesiveness, juiciness, and structural integrity in meat substitutes. Additionally, seaweed polysaccharides like sodium alginate are common texture enhancers and improve the structure of alternative meat and fish. Furthermore, microalgae’s bioactive compounds like polyphenols and antioxidants can improve product stability and shelf life, making algal proteins a valuable component in sustainable, clean-label formulations (Wu et al. 2023; Matos, Novelli, and Tribuzi 2022).
Achieving a meat-like texture with microalgae remains challenging, as high concentrations of algal proteins can reduce both firmness and elasticity depending on protein preparation and end product formulation (Espinosa-Ramírez et al. 2023). The algal cell wall, composed of approximately 10% cellulosic material and hemicellulosic components, contributes significantly to the rigidity of the cells, creating challenges for utilizing the algal biomass (Becker 2007). The presence of these complex components and polysaccharides in the cell walls of algae poses significant challenges in achieving a meat-like texture. The complex of cellulose, hemicellulose, and polysaccharide components interfere with protein network formation during processing, compromising the final products’ firmness (Bleakley and Hayes 2017). Optimized processing techniques, like high-moisture extrusion, texturizing, or mixing algae with soy or pea protein, are promising approaches for improving texture. Digestibility can be an additional challenge due to the robust cell walls of algae, which reduce protein bioavailability. Pretreatment methods, including enzymatic or physical disruption, may enhance digestibility and functionality (Espinosa-Ramírez et al. 2023).
Microalgae offer significant promise as a high-protein, sustainable resource for diverse food applications. While challenges related to texture, digestibility, and flavor persist, ongoing research into advanced cultivation and processing methods underlines their potential to benefit plant-based and alternative protein sectors. By leveraging these innovations, microalgae could play a crucial role in meeting global protein demand and advancing a more resilient, environmentally friendly food system.
Fermentation-derived ingredients and end products
FD proteins can be sourced from diverse microbes and similarly be used in a variety of applications, from meat, dairy, and egg substitutes to nutritional supplements, preservatives, colorants, texture regulators, and enzymes (Figure 3). This section highlights innovations in FD proteins enabling meat, egg, and dairy alternatives, exploring their applications, processes, and formulations.
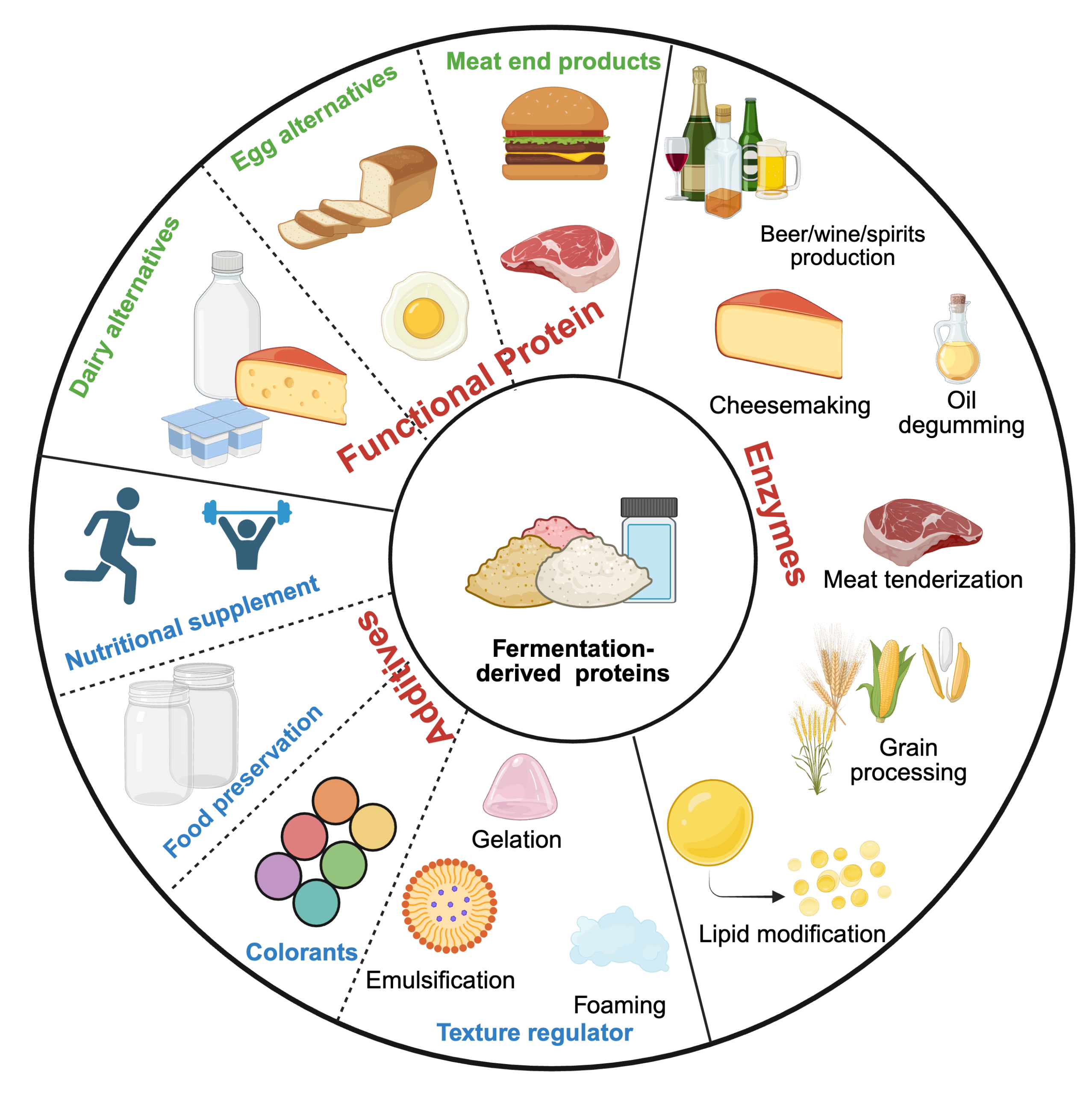
Figure 3. The diverse roles of fermentation-derived proteins and molecules in food functionality. Created with https://BioRender.com.
Fermentation-derived meat protein ingredients and products
Alternative meat products are primarily categorized into two main groups based on their structure:
- Whole cut products made of fibrils (Figure 4). These products are designed to replicate the fibrous structure and texture of whole cuts of meat, such as chicken breasts or steak.
- Restructured meat products (Figure 4), including coarse-particle products like ground/mincemeat and fine-particle (emulsion-based) products, such as gelled emulsions mimicking the smooth texture of hot dogs/frankfurters and deli meats. In fine-particle alternatives, achieving desired texture relies on gelation and forming a fine protein matrix, which eliminates the need for protein fibrillization (Xiong 2023).
To date, most examples of fermentation-derived biomass protein have come from fungi, thus more details are available regarding their applications and functionality as compared to microalgae and bacteria biomass protein ingredients. Commercialization of ingredients like mycoprotein began in the 1980s. These filamentous fungi-derived ingredients, along with other BD proteins, exhibit excellent gelling, emulsifying, and foaming properties, making them ideal for meat alternatives. Their filamentous structure and high protein content allow them to effectively mimic the textural properties of animal-derived proteins. Current fungal-based products include nuggets, burgers, fish fillets, and deli meats (Xiong 2023). Textured FD meat products can be developed using two primary approaches:
- Direct utilization of fungal biomass, producing microbial fermented meat.
- Processing isolated fungal proteins to simulate meat structure.
The production of fungal-based meats often involves combining mycoprotein with gel-forming agents such as acacia bean gum, agar, and alginate, and binding the aggregates using egg white protein or plant-based binders. Some biomass may contain endogenous molecules like polysaccharides that contribute to gelling without exogenous agents. These mixtures are heated to induce crosslinking, followed by cooling and freezing, which helps control ice crystal growth. This process binds filamentous mycelium into fibrous bundles, mimicking the texture of meat with a “fleshy” structure.
Modern techniques such as electrospinning, wet spinning, and 3D printing allow protein structuring at a molecular level, mimicking the intricate structure of animal meat (Figure 1). Additionally, larger-scale methods such as extrusion, freezing, shear cell technology, and hydrocolloid blending enhance the texture of fungal-based meat products by replicating animal-derived meat’s physical and structural properties. The combination of these techniques improves the overall quality and consumer appeal of meats (Figure 1; Figure 4) (B. Wang et al. 2023).
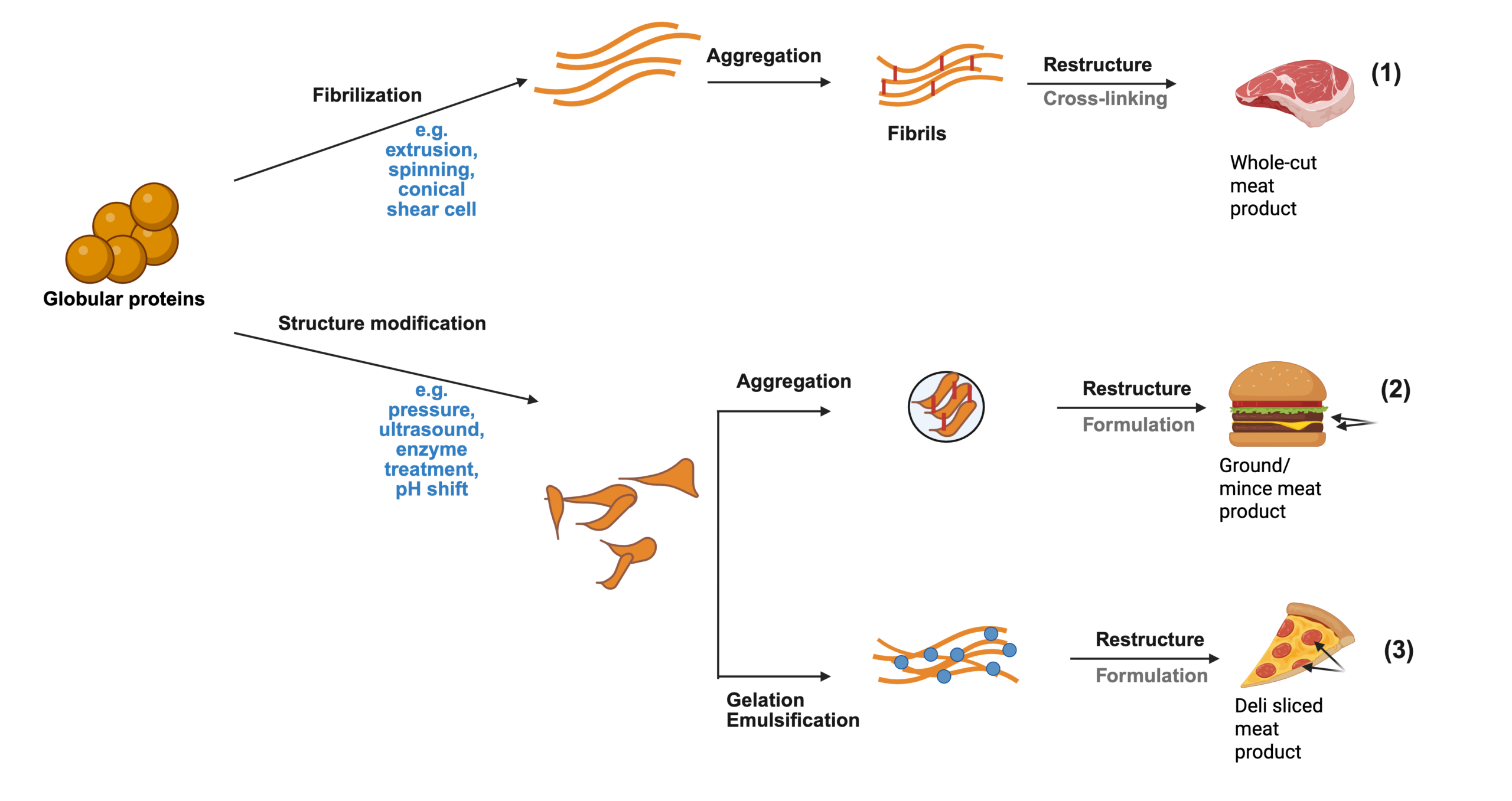
Figure 4. Production of meat alternatives from non-muscle proteins can be categorized into three types: 1: whole muscle-like structures, 2: coarse-particle products, and 3: fine-particle emulsion-based products (Xiong 2023). Created with https://BioRender.com.
Fungal biomass commercial end-products
The inherent structure of filamentous and mycelial fungal biomass has opened new avenues to its use in texturization and recreation of familiar end-products. For instance, combining Neurospora intermedia mycoprotein with plant-based excipients like soybean protein isolate and soluble starch results in well-textured and appealing end-products. Under optimized pH and salt concentrations, plus the addition of a soluble starch solution at 3%, these formulations balance elasticity, hardness, water retention, and fracture elongation. These organoleptic attributes are all key elements of texture acceptance. These findings demonstrate N. intermedia‘s potential for adoption as an AP meat ingredient (B. Wang et al. 2024).
Recent advancements in fungal strains and production technologies have led to the development of diverse fungal-based meat alternatives. Fermentation-derived protein producers leverage strains like Aspergillus sp., F. venenatum, and Pleurotus eryngii to create diverse commercially available products, including chicken substitutes, seafood, and bacon alternatives. As the industry grows, there is an expectation that new, food-safe microbes will be utilized to produce BD ingredients.
How is it made? A mycoprotein case study
Mycoprotein products made from F. venenatum biomass protein have been commercialized since the 1980s. In contrast to newly commercialized products or those scaling to commercial sale, many of the original patents on Quorn mycoprotein have expired, and the general process is well known. The mycelial fungi are grown and continuously harvested from airlift reactors in a fermentation that lasts up to 1000 hours (six weeks). The harvested biomass is heated to stop growth and to activate RNAse enzymes that reduce RNA to less than 2% of the biomass content (Moore et al. 2020). This is a requirement to reduce the purine content to regulatory limits for purines, which can be metabolized to uric acid, potentially leading to gout (Denny et al. 2008). After heating, the paste consists of fungal hyphae that contain 23–25 percent solids, the remainder of the mixture being water. The branching and length attributes are important for the meaty texture of mycoprotein; excessive branching can hinder the fibrous structure and increase the density of the biomass (Wiebe et al. 1992; Connell et al. 2025). Mycoprotein biomass, a viscoelastic paste, undergoes steaming, chilling, and freezing to achieve a texture similar to meat (Shahid et al. 2024). An example preparation process from mycoprotein paste is as follows (Figure 5) (Finnigan, Akintoye, and Mousavi 2016):
- Biomass Fermentation: F. venenatum fungi are grown, harvested from bioreactors, heated to stop growth and reduce RNA content, and dewatered into Mycoprotein paste that is ready for ingredient preparation and subsequent formulation
- Ingredient Mixing: Mycoprotein paste is mixed with additional ingredients to form a substantially homogeneous mass. These ingredients vary depending on the desired end product (e.g., meat-like pieces, mince, sausages, or roast meats).
- Forming and Steaming: The homogeneous mixture is passed through a forming machine to achieve the desired shape, then steamed at > 95°C for 35–45 minutes to initiate structural changes and partial cooking.
- Chilling: The steamed product is rapidly chilled to -5°C to -10°C for approximately 20 minutes. This step enhances the texture by making the product firmer and prepares it for subsequent processing.
- Optional Size Reduction: If necessary, the product undergoes size reduction by chopping/cutting to achieve specific dimensions or characteristics for particular applications like patties, nuggets, chunks, or mince/ground meat.
- Freezing and Texture Enhancement: A second texturization step is carried out by freezing the product. This step not only preserves the product but also contributes to the development of a firmer texture.
- Weighing and Packaging: The processed products are weighed and packaged according to desired retail or bulk specifications.
- Cold Storage: Packaged products are stored at -18°C for a minimum of seven days to allow the product to stabilize and achieve optimal quality before distribution.
- Distribution: Once ready, the product is delivered to retail outlets for sale to consumers.
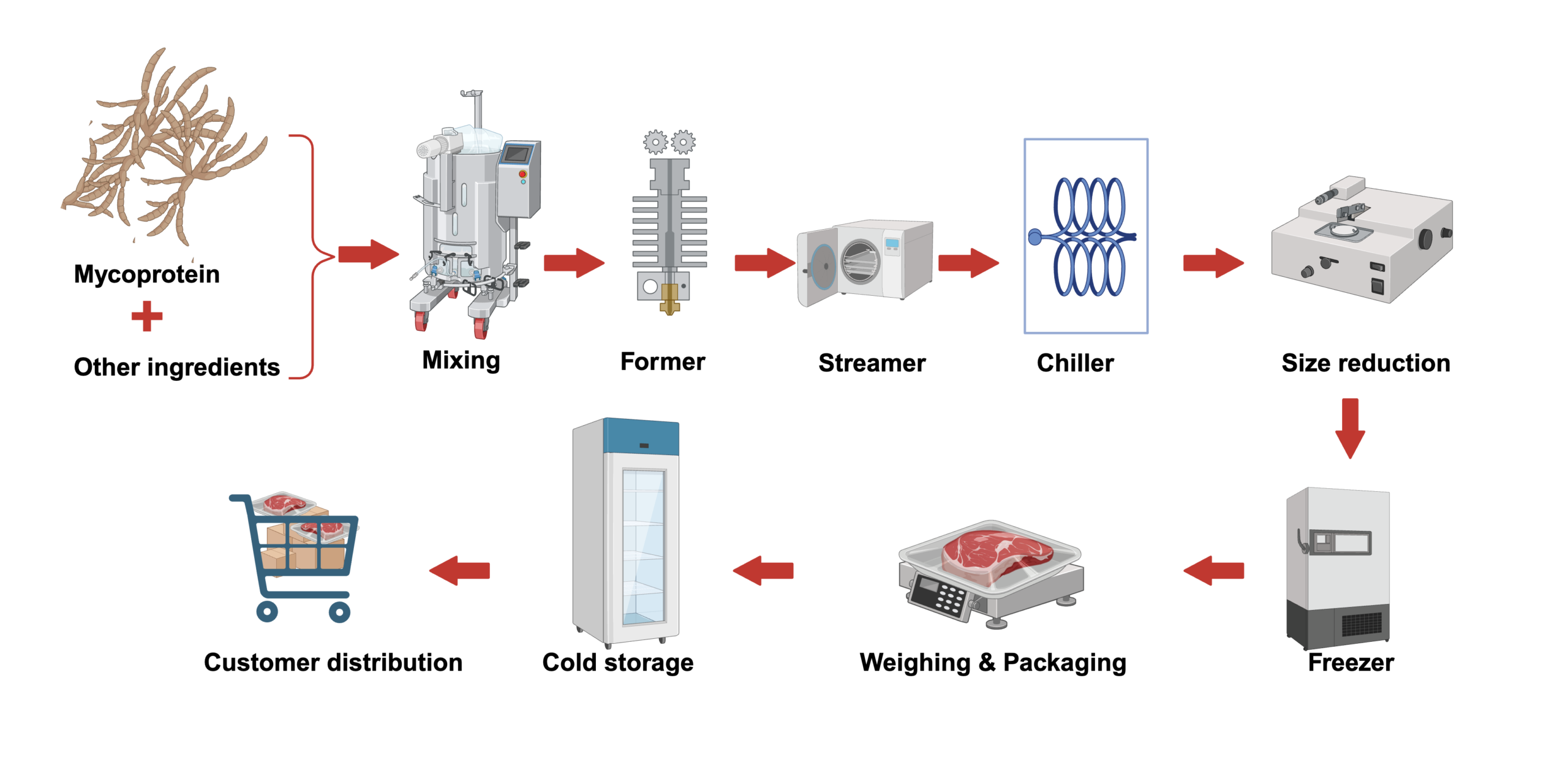
Figure 5. Production steps from “cradle to gate” for mycoprotein-based center of plate end-product. Created with https://BioRender.com.
As one example, the production of chicken-like mycoprotein could involve fermenting F. venenatum under optimized conditions in stirred tank or airlift reactors, which helps minimize shear stress on fungal mycelia. Reducing shearing forces preserves the fungal mycelial structures and ensures the desirable meat-like texture remains. After fermentation, the broth undergoes heating (to inactivate microbes and reduce RNA content), centrifugation, and dewatering to create a protein-rich biomass. The resulting biomass could be mixed with egg albumen (as a binding agent) or additional components, such as roasted barley malt, depending on the product type (e.g., mince, pieces, or nuggets). The mixture would then be shaped, steamed, frozen, and texturized to achieve a fibrous, meat-like texture (Figure 6). This example process would replicate chicken muscle fibers while producing a high-protein, sustainable product (Figure 6A/B) (Da Gama Ferreira, Misailidis, and Petrides 2023).
The blending of mycoprotein with other functional ingredients enables the end product to achieve favorable organoleptics and nutrition. Table 2 presents the formulation details of four different mycoprotein-based products from Quorn: Vegan Pieces, Vegan Mince, Vegan Burger, and Vegan Sausage. Each recipe includes a comprehensive list of ingredients along with their respective wet weight proportions (g/kg). These formulations are provided as representative examples to highlight the diversity in ingredient composition tailored to achieve specific textures, flavors, and functional characteristics suitable for various applications (Finnigan, Akintoye, and Mousavi 2016). Studies have also investigated innovative processing techniques such as high-moisture extrusion (HME) to modify the structure and digestibility of mycoprotein. Research revealed that untreated mycoprotein blends exhibit faster and higher amino acid absorption than HME-treated blends, emphasizing the need for careful optimization of processing parameters to preserve nutritional quality. These findings reveal challenges around the potential use of HME in mycoprotein ingredient preparation and justification for the freeze structuring utilized by some mycoprotein producers (West et al. 2024).
| Ingredients | Vegan Pieces (g/kg) | Vegan Mince (g/kg) | Vegan Burger (g/kg) | Vegan Sausage (g/kg) |
|---|---|---|---|---|
| Mycoprotein paste | 884.2 | 888.2 | 385.2 | 431.6 |
| Water | 12 | 0 | 171.1 | 170 |
| Potato protein | 32 | 50 | 32 | 32 |
| Vital wheat gluten | 10 | 10 | 10 | 10 |
| Calcium acetate | 4 | 4 | 3.9 | 4.4 |
| Calcium chloride solution | 12 | 12 | 3.9 | 4.4 |
| Sodium alginate | 0.8 | 0.8 | 0.8 | 0.8 |
| Carrageenan | 4 | 4 | 4 | 4 |
| Wheat fiber | 20 | 14 | 20 | 20 |
| Flavoring ingredients | 13 | 2 | 33 | 3 |
| Pea fiber | 8 | 6 | ||
| Onions | 100 | 62.5 | ||
| Meatless mince (ground mycoprotein) | 83.3 | 43.8 | ||
| Malt extract | 6 | 10 | ||
| Caramelized sugar | 9 | |||
| Texturized wheat protein | 83.3 | 37.5 | ||
| Oil | 20.5 | 60 | ||
| Flaked fat | 38.88 | |||
| Rusk | 68.8 | |||
| Tapioca starch | 10 | |||
| Seasoning | 31.2 |
Table 2. Formulation of Mycoprotein-Based Products: Vegan Pieces, Mince, Burger, and Sausage (Finnigan, Akintoye, and Mousavi 2016).

Figure 6. A) A prepared chicken-like mycoprotein product produced from Fusarium sp. biomass (photo courtesy of Quorn, used with permission). B) A prepared chicken-like mycoprotein product produced from Neurospora sp. Biomass (photo courtesy of The Better Meat Company, used with permission). C) Macroscopic images representing the color and visual structure of 20 wt% mycoprotein as a function of pH, ionic concentration, and salt, Image by Okeudo-Cogan et al. 2024 under the terms of the CC BY license. Created with https://BioRender.com.
Processing and formulation considerations for mycoprotein products
Mycoprotein has long been studied as a fermentation-derived biomass protein ingredient with potential for widespread commercial application. Work related to formulation and ingredient processing is of particular interest, especially in how it affects ingredient functionality in alternative meat products.
Processing to remove RNA must balance food safety with preserving product appeal. Thermal treatments to lower RNA content in microbial cells can partially denature proteins, affecting their functionality, while contributing to protein loss during ingredient preparation (Figure 1A). Enzymatic alternatives, such as the nuclease enzyme treatment, could effectively reduce RNA and DNA content without high heat exposure, thus preserving protein quality and concentration (US Food and Drug Administration, 2024). This PF-derived nuclease enzyme is an example of using a PF output as a processing aid for biomass fermentation-derived protein. Such enzymatic methods could offer an additional approach for the preparation and processing of BD protein ingredients.
In one study, replacing chicken breast meat with mycoprotein in breaded nuggets demonstrated texture properties (hardness, springiness, cohesiveness, or chewiness) comparable to conventional chicken nuggets. These nuggets exhibited lower lightness and yellowness color values but showed reduced cooking loss, ultimately achieving acceptable sensory and technological qualities (Hashempour-Baltork et al. 2023).
A key organoleptic benefit of mycoprotein is its ability to form textured, fibrous structures that resemble animal-derived meat products. However, binding is still required for final formulation into end products with the expected texture and mouthfeel. Typically, 3% egg white protein (EWP), primarily ovalbumin, is added to enhance gelation through disulfide bridges and improve the texture. A recent study explored mycoprotein (MYC)-EWP interactions, demonstrating that mycoprotein exhibited viscoelastic properties that were influenced by pH, salt type, and concentration. Acidic conditions increased storage modulus (G′), while calcium chloride (CaCl₂) had a greater impact than sodium chloride (NaCl) due to calcium crosslinking (Okeudo-Cogan et al. 2023). Imaging confirmed that EWP coated the fungal hyphae completely, moderating these effects. However, for price stability, allergenicity, and sustainability, it would be beneficial to identify alternatives to whole chicken EWP.
Studies exploring alternatives to EWP, such as potato protein (PoP) and multivalent cations (e.g., Fe³⁺ and Ca²⁺), revealed significant improvements in gel strength, texture, and nutritional quality (Okeudo-Cogan et al. 2024). Fe³⁺ influences fungal hyphae aggregation, particularly at lower pH levels, where it enhances protein-protein interactions but reduces composite strength by disrupting inter-hyphal binding. Conversely, CaCl₂ improves gel strength and texture, highlighting the dominant structuring role for calcium ions. When used together, Fe³⁺ and Ca²⁺ synergistically reduce excessive protein aggregation and enhance the overall structural stability of MYC-PoP composites. Interestingly, the effects of Fe³⁺ are pH-dependent, with acidic conditions promoting aggregation and weaker inter-hyphal interactions (Figure 6C). Therefore, these findings highlight the critical role of pH, ionic strength, and additive interactions in balancing protein aggregation and structural integrity. Moreover, this study demonstrates that PoP enhances fungal-protein composites by mediating electrostatic interactions, particularly in the presence of salts, positioning it as a promising allergen-free alternative to EWP (Okeudo-Cogan et al. 2024).
In addition to alternative chicken products, mycoprotein has shown remarkable potential in replacing traditional ingredients in sausages, nuggets, and patties. Studies replacing meat with mycoprotein in sausage formulations improved the nutritional profile of sausages by increasing essential amino acids (EAAs) and unsaturated fatty acids (UFAs). The closed nature of fermentation and the controlled environment in which the ingredient is processed may also reduce food contamination risks and increase microbial safety compared to meat sausages. Additionally, these sausages demonstrated superior water- and oil-binding capacities with potential for texture optimization (Shahbazpour et al. 2021).
Indeed, FD mycoproteins are a promising base ingredient for alternative meat formulations. Their potential for traditional center-of-plate product texture attributes, water holding capacity, and versatility makes them a large share of the biomass protein ingredients offered. However, the research space to find appropriate binders that can be used across mycoprotein products and the reduction of flavoring ingredients in some products (like the vegan burger, Table 2) highlight near-future food science and formulation advances that can further increase mycoprotein versatility and appeal.
Fermentation-derived alternative dairy ingredients
Alternative dairy products will become increasingly essential for sustainable and ethical food production. The growing demand for alternatives to livestock-derived dairy products has led to innovations in producing dairy-like proteins derived from fermentation and plant-based sources. A key challenge lies in replicating the nutritional profile, texture, and functional properties of dairy proteins, such as caseins and whey, while ensuring environmental sustainability and catering to the preferences of health-conscious and ethical consumers.
Milk contains approximately 87–89% water, with the remaining fraction consisting of proteins (3.0–3.9%), fats (3.3–5.4%), and sugars (4.4–5.6%), and trace amounts of salts and vitamins. The protein fraction consists of 80% caseins and 20% whey proteins, making it highly nutritious due to its complete essential amino acid profile. Whey proteins, including β-lactoglobulin (β-LG, 52%), α-lactalbumin (α-LA, 17%), glycomacropeptides (12%), immunoglobulins (10%), serum albumin (5%), and lactoferrin (1.5%), are used in various applications such as infant formula, yogurt, cheese, and sports nutrition products. β-LG, a key bovine whey protein, is nutritionally valuable despite being absent in human milk. Caseins, including β-casein (36%), κ-casein (14%), α-s1-casein (40%), and α-s2-casein (10%), are heat-stable proteins with unique functional properties such as emulsification and water-binding, making them particularly suitable for coffee whiteners and other heated products (Nielsen, Meyer, and Arnau 2024).
The recombinant production of casein and whey proteins has made remarkable strides through microbial fermentation. Microbial hosts like K. phaffii, S. cerevisiae, and E. coli produce recombinant bovine κ-casein and other dairy proteins, with significant progress achieved in the yield and efficiency of recombinant protein production (Eastham and Leman 2024). For instance, companies are utilizing PF to scale up to commercial production of bovine β-casein for animal-free cheese alternatives, replicating the texture, flavor, and melting profile of mozzarella. Similarly, whey proteins like α-lactalbumin and β-lactoglobulin are efficiently produced in microbial systems, enabling sustainable dairy alternatives (Iglesias-Figueroa et al. 2016; Kim et al. 2007).
Recombinant dairy proteins, such as β-lactoglobulin and α-lactalbumin, are increasingly being used in the dairy industry due to their excellent functional properties, including emulsification, foaming, and heat stability (Diaz-Bustamante et al. 2023; Hoppenreijs et al. 2024). FD production and subsequent isolation of recombinant dairy proteins produce a homogenous white powder suitable for various food applications (Diaz-Bustamante et al. 2023). However, these proteins are rarely used in isolation. They are marketed to food manufacturers, sold in bulk, and used as ingredients for various alternative dairy applications. Often, formulators integrate recombinant dairy proteins with plant-based fats, carbohydrates, salts, vitamins, calcium, and minerals to create cheese alternatives (Diaz-Bustamante et al. 2023). These proteins are produced through PF using microorganisms like Trichoderma reesei, Pichia pastoris, and K. lactis, which are capable of producing commercial titers (~15g/L and higher) of recombinant proteins with the required post-translational modifications (Diaz-Bustamante et al. 2023).
PF has become a key technology for producing recombinant milk proteins. Companies like Perfect Day employ Trichoderma reesei to produce β-lactoglobulin, the first commercial example of a PF whey protein for products such as ice cream and alternative milk products (Figure 7A). Other companies, including New Culture and Change Foods, focus on producing recombinant caseins using PF technologies (Nielsen, Meyer, and Arnau 2024).
Whey proteins are proportionately the largest protein fraction of milk and contribute to many of the functional attributes of dairy products. In bovine milk, β-lactoglobulin is the major whey protein; therefore, there is a high interest in producing the protein using PF approaches. At research scale, this protein was expressed from engineered T. reesei fungi, purified, and tested to demonstrate functionality comparable to its cow milk-derived counterpart (Aro et al. 2023). Bovine β-lactoglobulin exhibited exceptional emulsification properties, forming stable emulsions without phase separation for 30 days at 4°C, making it ideal for long-shelf-life food products (Figure 7B).
While recombinant whey production has seen earlier development and commercialization, various companies and startups are now turning to casein. Progress on PF-derived caseins has been slower, largely due to their more complex interactions with calcium salts and fats (Diaz-Bustamante et al. 2023).
Nevertheless, the production of casein proteins through biotechnology also holds great potential. Reconstituting casein protein with calcium and fats produces the micelle structures that agglomerate into cheese curds, a process that is crucial for traditional cheese-making. To date, the ideal peptide length, post-translational modifications, and downstream processing pathways to create a PF casein ingredient, and subsequent micelle formation, are still under investigation (Chezan et al 2026). For example, the phosphorylation of casein appears to play a role in its interaction with fat and calcium, and fully dephosphorylated recombinant casein fails to properly fall out of solution and interact with calcium (Che et al. 2025). Indeed, recombinant phosphomimetic casein protein, where serine amino acids are replaced by aspartic acid in the protein sequence, were able to achieve casein micelle formation, paving the way for additional avenues towards casein functionality and micelle formation (Balasubramanian et al. 2025).
Attempting to recreate the exact biochemistry of cheese curds may not be required for successful PF-casein containing cheese formulations. Recently, patent filings have disclosed non-micelle-based methods for achieving casein functionality in alternative cheeses (Radman 2023). Using these biotechnologically produced caseins, PF-enabled cheese typically requires blending PF-derived casein with water, oils, and small quantities of carbohydrates. Emulsifiers, stabilizers, and/or flavoring agents are also potential ingredients, depending on the type of cheese produced. The mixture is subjected to heating and mechanical processing to achieve a texture, meltability, and stretchability similar to traditional cheese (Figure 7C). The final product is shaped into various forms, such as blocks, slices, or shreds with additional treatments to improve its sensory properties. These cheese analogues exhibit a smooth and cohesive texture, excellent melting and stretchability, and a customizable flavor profile that closely mimics traditional dairy cheese. Casein proteins contribute essential functionality to cheese with the ability to bind to fats (melting), induce increased binding when cooked (stretching), and ionic attraction of salts (taste). This innovative approach provides a sustainable and versatile alternative to traditional cheese production (Radman 2023).
An example innovation in plant-based dairy alternatives is the implementation of concentrated microalgae flour to produce lactose-free milk with a nutritional profile comparable to cow’s milk, including high-quality essential amino acids (Wu et al. 2023). Unlike previous approaches that used algae as supplements or additives, current efforts focus on a fully algae-based dairy substitute derived from protein-rich microalgal flour (Cornall 2021; Ho 2021). By adjusting the ratio of water-soluble microalgae flour, formulators can customize protein content and enhance sensory properties, paving the way for a range of alternative dairy products like yogurts, cheeses, and ice creams.Ice cream is an example of a commercial application opportunity for fermentation-derived protein ingredients. An FD protein ingredient with plant-based fats and sugars can form the basis of an animal-free ice cream. In some cases, these proteins can be biomass-derived, such as in the case of the collaboration between the Danish Technological Institute and Sophie’s Bionutrients, which developed a Chlorella microalgae-based ice cream with a higher protein content than many plant-based alternative ice creams (Figure 7D) (Southey 2022). Similarly, PF-derived beta-lactoglobulin protein adds protein content and creamy texture to ice cream (Smith 2024). In both examples, FD protein ingredients drive organoleptic improvement of animal dairy-free desserts while enabling lactose-free formulations.

Figure 7. A) A whipped alternative dairy product with PF β-lactoglobulin to enhance foaming (photo courtesy of Perfect Day, used with permission); B) Left: Glass vial of PF-derived bovine β-lactoglobulin and right: Glass vial of control animal-derived β-lactoglobulin were stored undisturbed at 4 °C for 30 days (Aro et al. 2023 under the terms of the CC BY license); C) Mozzarella cheese using PF casein protein demonstrating stretching and melting (photo courtesy of New Culture, used with permission); D) An ice cream product using microalgae-derived protein as an ingredient, demonstrating creaminess in an animal dairy-free formulation (photo courtesy of Sophie’s Bionutrients, used with permission); E) A whipped egg ingredient using PF ovalbumin demonstrating foaming (Bioalbumen®, photo courtesy of Onego Bio, used with permission); F) A bread baked using PF ovalbumin demonstrating binding in baked goods (Bioalbumen®, photo courtesy of Onego Bio, used with permission). Created with https://BioRender.com.
Fermentation-derived alternative egg protein ingredients and products
PF offers a promising approach to replicate the functional and nutritional properties of eggs without animal-derived components. Raw eggs consist of 12.5–13% protein, with ovalbumin comprising the majority (54%) of egg white proteins. Other key proteins include ovotransferrin (12%), ovomucoid (11%), and smaller amounts of ovomucin and lysozyme (each 3.5%). These proteins exhibit desirable functional properties, such as foaming, emulsifying, and gelling, making them invaluable in cheese production, confectionery, and baking (Nielsen, Meyer, and Arnau 2024).
Egg whites also contain biologically active proteins with various functional benefits. Lysozyme, for example, is used as a food preservative due to its antimicrobial properties, ovotransferrin functions as a metal transport agent with antimicrobial and anticancer effects, and ovomucin and ovomucoid exhibit tumor-suppressive properties. These qualities emphasize the importance of eggs in global diets and industrial applications (Knychala et al. 2024).
Egg ingredient alternatives have gained significant attention due to animal welfare, sustainability, and global food security concerns. Eggs are valued for their high nutritional content and their functional properties, such as coagulating, emulsifying, foaming, and coloring. However, the contribution of animal-based foods to global carbon emissions, poultry diseases reducing egg supply, and unstable pricing have driven consumer and industry interest in egg alternatives. As a result, the global egg alternative ingredient market has a projected 2025 value of $1.6 billion US dollars (Jin, Seo, and Kim 2024).
PF egg white proteins offer several advantages, including scalability, reproducibility, and absence of contaminants such as pathogens commonly associated with egg proteins. In isolation, it also enables the production of specific egg white proteins, such as ovalbumin, lysozyme, and ovotransferrin, enhancing their functionality for targeted applications. The PF-derived proteins closely replicate the functional properties of egg proteins across various food applications. Patent filings demonstrate various functional claims for PF-derived ovalbumin (Mahadevan et al. 2021). For example, PF-derived ovalbumin provides excellent foaming and emulsification, ensuring light textures in baked goods (Figure 7E). The foaming properties are especially useful in products like cakes and meringues, where stable foam structures are essential for texture and volume (Figure 7F). Recombinant ovalbumin stabilizes and emulsifies in mayonnaise and dressings, maintaining smooth textures and sensory qualities in animal-free formulations. It also enhances binding and gelation in plant-based meat alternatives, improving texture and mouthfeel to mimic animal-based counterparts. Fine-tuning ovalbumin’s gelling properties through temperature, pH, and salt can create specific textures in alternative meats, offering versatility in formulation. Ovalbumin’s gelling ensures stability, creaminess, and a premium texture in dairy-free desserts like mousses and ice creams. Finally, recombinant ovalbumin is a high-quality protein in nutritional supplements and protein powders, offering allergen-free, clean-label solutions for health-conscious consumers (Mahadevan et al. 2021).
Recent advancements in PF have enabled the production of ovalbumin and other egg proteins in microorganisms (Nielsen, Meyer, and Arnau 2024). Recombinant production of egg white proteins, particularly ovalbumin, has been explored since the 1970s using hosts such as E. coli, S. cerevisiae, and K. phaffii. While initial attempts yielded minimal production, recent advancements with hosts like K. phaffii have enabled production levels of 5.45 g/L for ovalbumin, 0.1 g/L for chicken ovotransferrin, and smaller proteins like lysozyme (0.4 g/L) and avidin (0.33 g/L) (Knychala et al. 2024). Recent commercial breakthroughs include the production of ovalbumin in Trichoderma reesei, in Aspergillus niger, and Komagataella phaffii (Nielsen, Meyer, and Arnau 2024). Companies have made strides in producing EWP at high titers, with Every Company reporting ovalbumin titers from 17-30 g/L in K. phaffii and Onego-Bio reporting 120 g/L in T. reesei, which are significant strides compared to the literature published titers in S. cerevisiae (Jin, Seo, and Kim 2024), K. phaffii (Yang et al. 2009), andT. reesei (Aro et al. 2023) for EWPs. These developments highlight progress on commercial scalability and reproducibility of PF for egg protein production, signaling their near-term availability for commercial food production.
Further, these proteins provide functionalities higher than or equivalent to animal-derived egg whites (Knychala et al. 2024). Patents in this area emphasize producing innovative recombinant egg-white protein compositions. For example, methods for expressing two or more egg-white proteins through PF and producing recombinant ovalbumin for egg-free food formulations have been filed for IP protection (Anchel 2016; Mahadevan et al. 2021). PF companies have patent-protected innovative solutions on highly soluble egg-white proteins and egg white proteins from other bird egg species with enhanced functionality, like gelation over traditional hen egg ovalbumin.
Health and nutrition of fermentation-derived ingredients
This section explores the nutritional profile, health benefits, and potential applications of these innovative protein sources, emphasizing their roles in alternative meat, egg, and dairy production.
Microbial biomass is a rich source of protein, lipids, and essential vitamins, particularly B vitamins such as biotin, folic acid, niacin, pantothenic acid, pyridoxine, riboflavin, and thiamine. However, only certain microbes, bacteria, and archaea produce vitamin B12, an essential cofactor for DNA synthesis and metabolism (Fang et al. 2017). Cyanobacteria microalgae, such as Chlorella, are reliable sources of active vitamin B12, while Spirulina predominantly contains pseudovitamin B12, which is inactive in humans (van den Oever and Mayer 2022). In designing SCP ingredients, using microalgae and cyanobacteria can coproduce vitamin B12 and other B vitamins along with proteins (Linder 2019).
Microbial sources like microalgae and cyanobacteria also provide other critical nutrients, including vitamins C and E (Del Mondo et al. 2020). Additionally, certain microorganisms synthesize carotenoid pigments like β-carotene, a precursor to vitamin A, and astaxanthin, responsible for the orange and red hues in crustacean shells and salmon meat (Barreto et al. 2023; Agarwal et al. 2023). Oleaginous microorganisms can accumulate 50–70% lipids in their dry biomass under nitrogen-limited conditions, yielding single-cell oils rich in polyunsaturated fatty acids like omega-3 fatty acids (Linder 2019).
The carbohydrate composition of microbial biomass varies by species, influencing its nutritional and functional applications. These carbohydrates can be either digestible nutrients, providing energy, or indigestible fibers, which play an essential role in supporting gut health and maintaining a balanced diet. For example, cellulose and pectin found in eukaryotic microalgae contribute to dietary fiber intake (Niccolai et al. 2019), while β-glucan and chitin in fungi have been shown to promote gut microbiota diversity and support improved glycemic control (Finnegan et al. 2019). Eukaryotic microalgae contain cellulose, pectin, mannan, or xylan, while bacterial cell walls contain peptidoglycan. Fungi like yeasts and filamentous fungi possess cell walls enriched with β-glucan and chitin, contributing to their functional and health-related properties, including potential gut health benefits and glycemic homeostasis – the regulation of blood glucose levels (Linder 2019). Mycoprotein consumption has been shown to improve insulin sensitivity and reduce blood cholesterol levels, particularly in individuals with elevated baseline cholesterol (Pavis et al. 2024). Production processes reduce mycoprotein’s initial RNA content (10%) to under 2%, mitigating health risks like gout. Research is ongoing to optimize its sensory and health properties, broadening the product selection and increasing accessibility to diverse consumer groups (Souza Filho et al. 2019).
The nutritional value of filamentous fungi highlights their potential as a sustainable protein source. While fungi such as F. venenatum exhibit amino acid profiles and digestibility comparable to animal-derived proteins, the chitin-rich cell walls of fungi can slightly hinder digestibility. Complementing this analysis, researchers have analyzed the in vitro protein digestibility and amino acid accessibility of five fungal species, such as A. oryzae, N. intermedia, F. venenatum, Rhizopus delemar, and Rhizopus oligosporus, comparing them to casein, salmon, chicken, and beef proteins. The degree of protein hydrolysis for fungi ranged between 43% and 58%, slightly lower than that of salmon, chicken, and beef (59%–67%). Notably, A. oryzae and R. oligosporus displayed amino acid accessibility and essential amino acid profiles comparable to muscle proteins (R. Wang et al. 2023). Amino acid accessibility during digestion can vary depending on microbial species, with F. graminearum at 78% and F. venenatum at 86% amino acid accessibility. Compared to milk protein (95%) and animal meat digestibility (90%-95%), there is room for increasing amino acid access, though it should be noted that amino acid accessibility is similar to egg white protein at 91% (Evenepoel et al. 1998; R. Wang et al. 2023 ). Preprocessing techniques like freeze-drying and milling significantly enhanced digestibility, particularly in A. oryzae biomass. Future approaches for downstream processing and ingredient preparation may help increase digestibility and amino acid accessibility for microbial biomass protein.
Food safety and regulatory considerations of fermentation-derived ingredients
The global growth of the alternative protein sector depends on establishing clear, consistent, and harmonized regulatory frameworks. Such frameworks ensure consumer safety and facilitate international trade and market access. This section examines the regulatory landscape and safety challenges of FD proteins, emphasizing the importance of transparency and innovation in fostering consumer trust.
The global progress of alternative proteins in the food industry can be incremental due to the lack of harmonized national and international regulation frameworks that govern novel foods. Alternative proteins are typically categorized as novel foods, defined as foods or ingredients without a history of safe use, though regulatory approaches vary globally. Check out GFI APAC’s Novel Food Regulations Around the World website for a more detailed analysis. In the European Union, novel foods are regulated under the European Commission’s 2015/2283 legislation, effective from 2018. Conversely, the United States lacks a specific definition or regulation for novel foods. The U.S. Food and Drug Administration (FDA) treats new food ingredients as either food additives requiring premarket approval or to be evaluated and greenlit for food use as generally recognized as safe (GRAS). In Asia, regulatory frameworks for novel foods are less developed than in Europe or the U.S., with only a few countries, such as China, South Korea, Singapore, and Thailand, having specific regulations. China follows a model similar to that of the EU. At the same time, South Korea has temporary standards for novel ingredients, including recent provisions allowing plant-based meat alternatives to use the term “meat” on labels. Singapore and Thailand, however, have more comprehensive safety assessment guidelines. Singapore requires safety evaluations of novel ingredients under international standards, while Thailand mandates clinical or sub-chronic studies to ensure consumer safety. In Europe, the European Food Safety Authority (EFSA) evaluates the safety of novel foods, including PF-derived products, before issuing regulatory approvals. While EFSA has yet to approve PF protein food products, several are under review. Strains of microbial species like Bacillus subtilis, Saccharomyces cerevisiase, Chlamydomonas reinhardtii, and Corynebacterium glutamicum have a long history of safe use in food production (EFSA Panel on Biological Hazards). International organizations such as the Food and Agriculture Organization (FAO), World Health Organization (WHO), FDA, and EFSA recognize these strains as safe, making them suitable candidates as host microorganisms for PF applications (Mefleh and Darwish 2024).
Across jurisdictions, there are separate considerations for genetically modified organisms, which cover many of the host organisms used to produce precision FD ingredients, even if these organisms are not present in the final product (Hanlon and Sewalt 2020). These regulatory differences highlight the need for global cooperation and harmonized guidelines to facilitate the growth of the alternative protein industry (Malila et al. 2024).
PF-derived meat, dairy, and egg proteins are a rapidly growing food category. In the U.S., regulatory greenlighting of specific proteins, such as β-lactoglobulin (β-LG) from Perfect Day and ovomucoid from The EVERY Company, has led to their commercial sale and use as a food ingredient. One key issue is determining appropriate usage levels in food products to ensure safety and functionality. Another challenge is enhancing consumer awareness of the ingredients produced by PF. If consumers are allergic to animal-derived dairy or egg proteins, it’s highly likely they will be allergic to the PF-derived replacement protein. Therefore, unambiguous identification and labeling of these novel ingredients is necessary to enhance consumer awareness of this equivalence and potential allergenicity. Harmonizing regulations and improving transparency are vital to increasing consumer confidence and supporting the broader adoption of PF proteins in the food industry (Nielsen, Meyer, and Arnau 2024).
The safety evaluation of PF products focuses on the potential toxicity and allergenicity of the host microbe, its accompanying (copurifying) molecules, and the food ingredient itself. PF-derived proteins may introduce new allergens or alter the allergenic properties of the product due to changes in protein sequence. Comprehensive safety testing is required to mitigate these risks, including analyzing amino acid sequences for similarities to known allergens (Goto et al. 2023; Grundy et al. 2024). For instance, the FDA approved yeast-produced β-lactoglobulin for use in plant-based foods (excluding infant milk) only after rigorous safety assessments, including scrutinizing the product for potential allergens (US Food and Drug Administration, 2020). In biomass fermentation, rare adverse reactions to mycoprotein products have been reported in individuals with mold allergies (Katona and Kaminski 2003). Therefore, mycoprotein products carry labels with precise wording describing the product as potentially allergenic for those with mold allergies.
The potential production of toxic metabolites by microbes and contamination with undesirable microorganisms are concerns that must be scrutinized and derisked during product development. Mycotoxins are toxic substances produced by a subset of filamentous fungi under specific conditions, pose a potential hazard, and can accumulate in tissues. Many foods have a risk of mycotoxin accumulation due to fungal contamination, such as soil-grown tubers, legumes, grains, and dried fruit. Improper processing or storage can lead to mycotoxin exposure, which poses a high risk to sensitive populations such as pregnant people, children, geriatric populations, and immunocompromised individuals. Utilizing sidestreams or feedstocks where molds have propagated requires extra care and surveillance to avoid transferring mycotoxins to yeast biomass. Strict processing protocols and safety measures, employed during GMP (“good manufacturing practices”) food production, are essential to mitigate these risks (Jach et al. 2022). Therefore, these mycotoxins are routinely tested for, and demonstrating their absence is typically considered a regulatory hurdle that must be met. Additionally, regulators often require the genetic sequences of microbes to be analyzed and the identification of any known mycotoxin-producing genes to be declared. For example, while some strains of F. venenatum can produce mycotoxins, the strains used commercially by Quorn do not make these toxins (O’Donnell, Cigelnik, and Casper 2018). Continued scrutiny and surveillance of mycotoxins, common in the food industry, is also required in fermentation settings, especially when investigating novel microbes and production conditions (Malila et al. 2024).
International cooperation among regulatory agencies is crucial to advance the alternative protein sector. Establishing globally accepted safety and quality standards would reduce trade barriers and ensure a level playing field for producers. Research into advanced safety evaluation techniques, such as proteomic and metabolomic profiling, could enhance the identification of potential allergens or toxins, providing more comprehensive safety assessments. Public engagement and education are equally important for addressing consumer concerns regarding the safety, sustainability, and nutritional value of FD proteins.
By fostering trust, harmonizing regulations, and supporting ongoing innovation, the alternative protein industry can overcome its current challenges and pave the way for widespread acceptance and adoption of these innovative food solutions.
